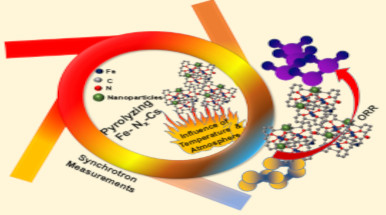
The depletion of fossil fuels and ongoing ecological challenges underscore the pressing need for sustainable and environmentally friendly energy solutions. In this pursuit, fuel cells have become a focal point in current research, primarily due to their exceptional advantages, with the noteworthy aspect being the recognition of hydrogen as a green energy carrier. However, the biggest obstacle in the mass-scale commercialization of fuel cells is the utilization of scarce and overpriced platinum-based (PGMs) cathode side electrocatalysts to carry out sluggish and complex oxygen reduction reactions (ORR). Over the past few decades, transition metal-nitrogen-carbons (TM-NX-C) have been developed and demonstrated a remarkable potential to replace PGMs for advanced electrocatalytic applications. Among the first-row transition metals, iron is considered the metal of choice to fabricate Fe-Nx-C electrocatalysts for ORR with appreciable kinetics due to its suitable thermodynamic interaction with oxygen and ability to launch tetra-electronic reduction of oxygen, particularly when the iron is present in close coordination with nitrogen (Fe-Nx). To evolve and engineer a robust active-site structure of Fe-Nx-C with multitudes of functional moieties, pyrolysis is employed as the key fabrication methodology. Nevertheless being the most employed fabrication methodology, pyrolysis itself remains the least understood phenomenon and therefore can be regarded as a Blackbox. From the core knowledge of materials science, it is the processing technique that dictates the evolution of structure and hence ultimately decides the performance during application. Therefore, the understanding of the evolution and transformation of active sites over the course of pyrolysis is a crucial inquiry.
The research team of the University of Milano-Bicocca (Mr. Mohsin Muhyuddin, Mr. Seyed Ariana Mirshokraee, Dr. Jacopo Orsilli, Prof. Roberto Lorenzi) led by Dr. Carlo Santoro of the Department of Materials Science, in collaboration with Prof. Plamen Atanassov, Dr. Eamonn Murphy and Dr. Shengyuan Guo at the University of California, Irvine, USA, Dr. Enrico Berretti, Dr. Laura Capozzoli and Dr. Alessandro Lavacchi at CNR-ICCOM, Florence, Italy and Dr. Francesco D’Acapito at CNR-IOM (beamline BM08 ESRF) Grenoble, France, initiated a comprehensive study to examine the structural evolution of active sites in Fe-Nx-C during pyrolysis. To address uncertainties in the fabrication of electrocatalysts during pyrolysis, this investigation utilized a combination of advanced in-situ and ex-situ characterization tools. The analysis focused on examining the impact of pyrolysis conditions on the structural evolution of the Fe-Nx-C electrocatalysts and making a relationship among pyrolysis parameters, structure and electrochemical performance. In a nutshell, high surface area carbon black was functionalized with iron phthalocyanine and pyrolyzed from 25 ºC to 1000 ºC in two different atmospheres i.e. pure Ar and slightly reducing Ar/H2. Sample fabrication at each temperature and in both atmospheres was thoroughly elucidated in in-situ and ex-situ synchrotron X-ray absorption spectroscopy (XAS) at ESRF Grenoble, France. To define the trends in surface chemistry, a thorough X-ray photoelectron spectroscopic (XPS) analysis was conducted. Detailed high-resolution transmission electron microscopy (HR-TEM) of the samples was carried out. The ORR electrocatalysis was performed in both acidic and alkaline media to specify the role of active-site structure evolved under different pyrolysis conditions. The findings of this study have been documented in the manuscript. No doubt, the in-situ characterizations, coupled with ex-situ surface, microscopic and spectroscopic analysis, along with electrocatalytic measurements, are crucial for advancing the next-generation ORR electrocatalysts for sustainable energy production via fuel cells. The authors are confident that this approach can be readily applied to other electrochemical reactions and various types of electrocatalysts derived from single atoms or nanoparticles.
The results of the research are reported in the paper “Formation of the active site structures during pyrolysis transformation of Fe-phthalocyanine into Fe-Nx-C electrocatalysts for the oxygen reduction reaction” (DOI: 10.1016/j.apcatb.2023.123515) published in Applied Catalysis B: Environmental (Impact Factor 22.1, Journal Citation Report (Clarivate Analytics, 2022)).